Oligomerix Mission:
Oligomerix seeks to improve the lives of patients with neurodegenerative diseases by discovering and developing a portfolio of disease-modifying, oral small molecule therapeutics that are simple to use.
Company Overview:
Oligomerix is a biotech company that has developed a proprietary platform technology for drug discovery and biomarker development for central nervous system (CNS) disorders focused on neurodegeneration and dementias, and has discovered unique, highly differentiated drug-like small molecules that inhibit protein self-association into toxic oligomers.
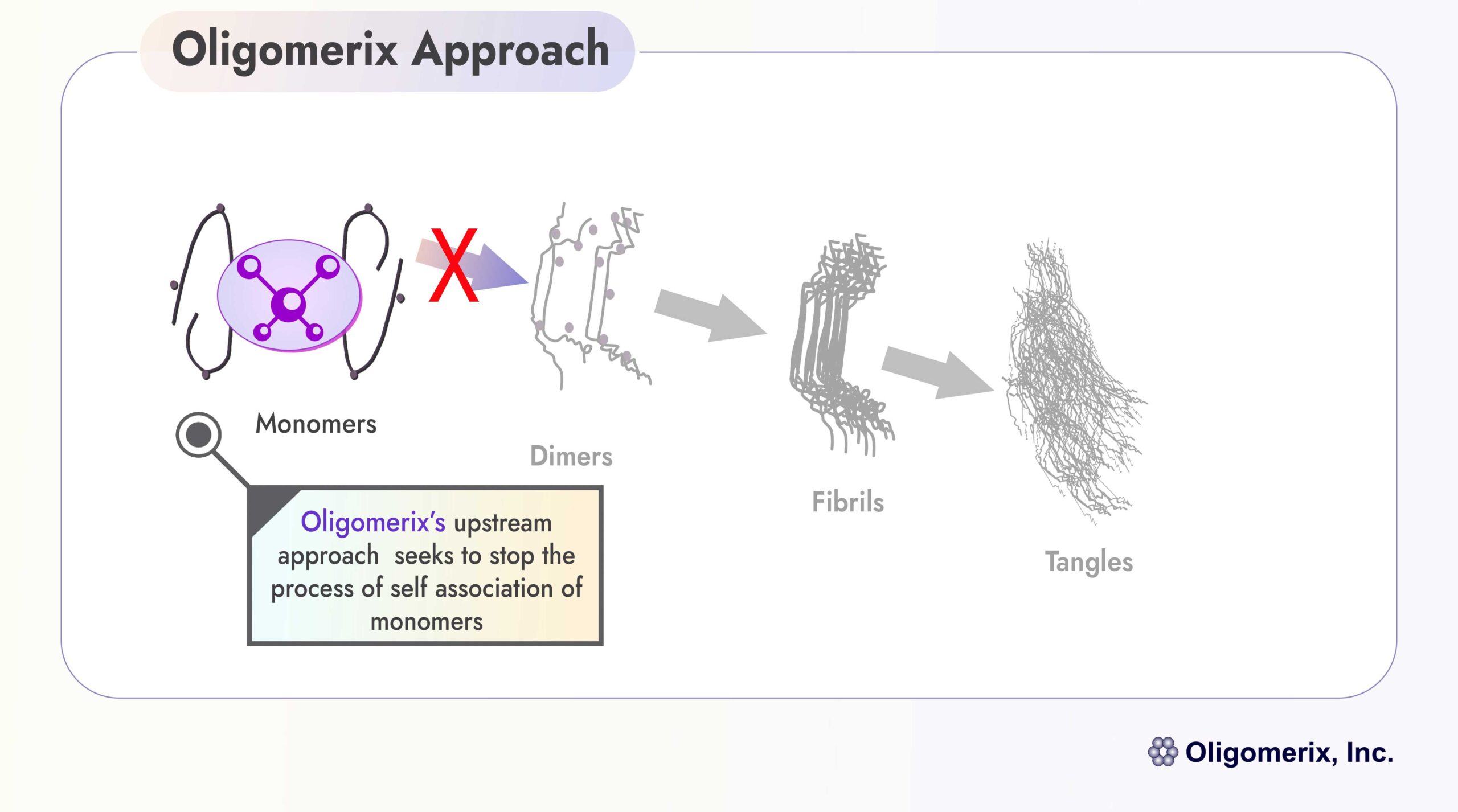
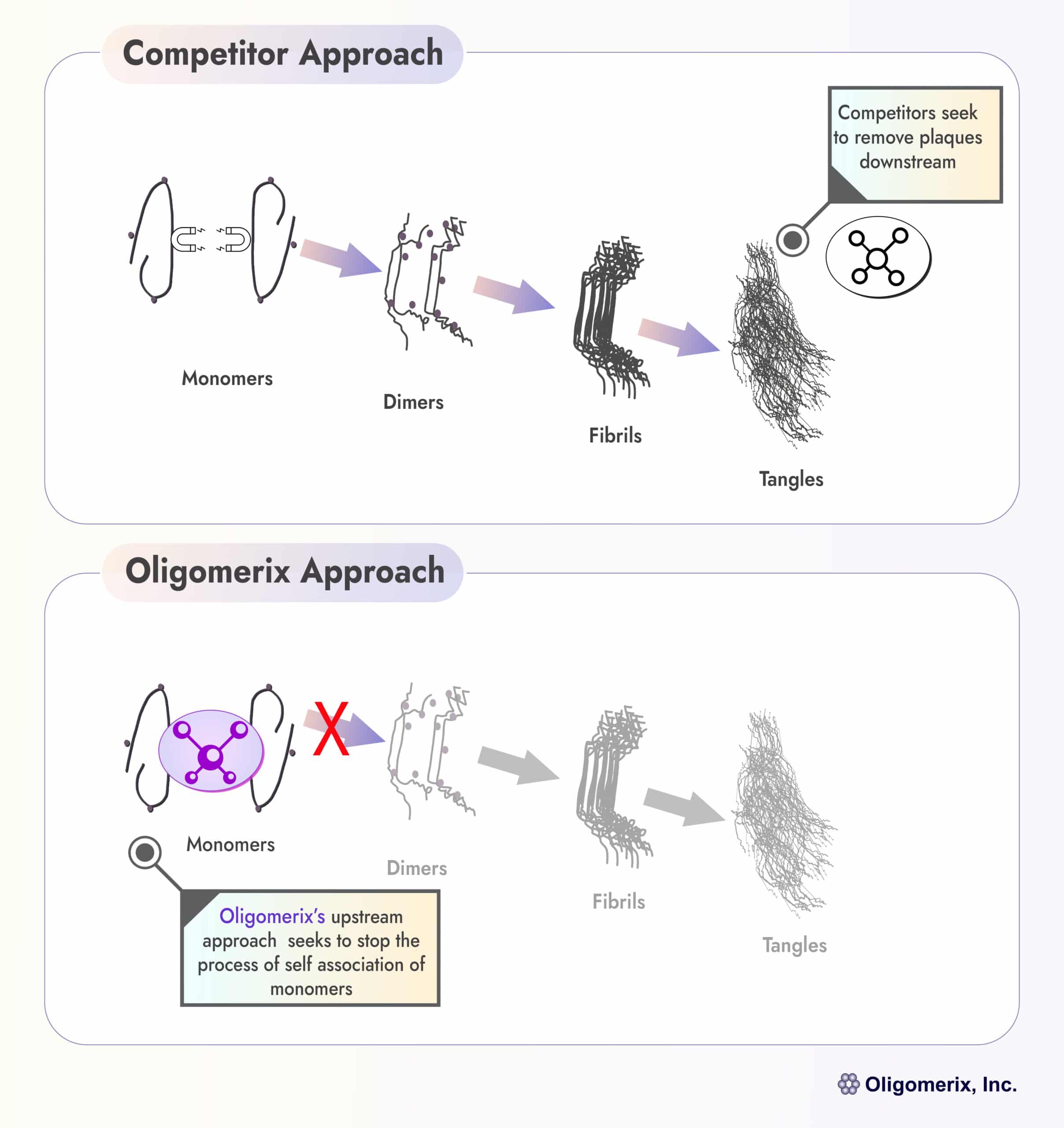
Oligomerix targets the beginning of the tau protein aggregation cascade to minimize the formation of toxic aggregates and downstream tangle formation - thereby preventing neurodegenerative disease progression.
An effective inhibitor of tau self-association has potential utility in the treatment of various tauopathies including PSP, FTD and AD, because it inhibits the formation and growth of all pathological multimeric tau structures associated with these diseases.
Oligomerix has positioned itself to be the premier provider of highly differentiated small molecule therapeutic interventions for blocking the formation of toxic tau oligomers and higher order aggregates.
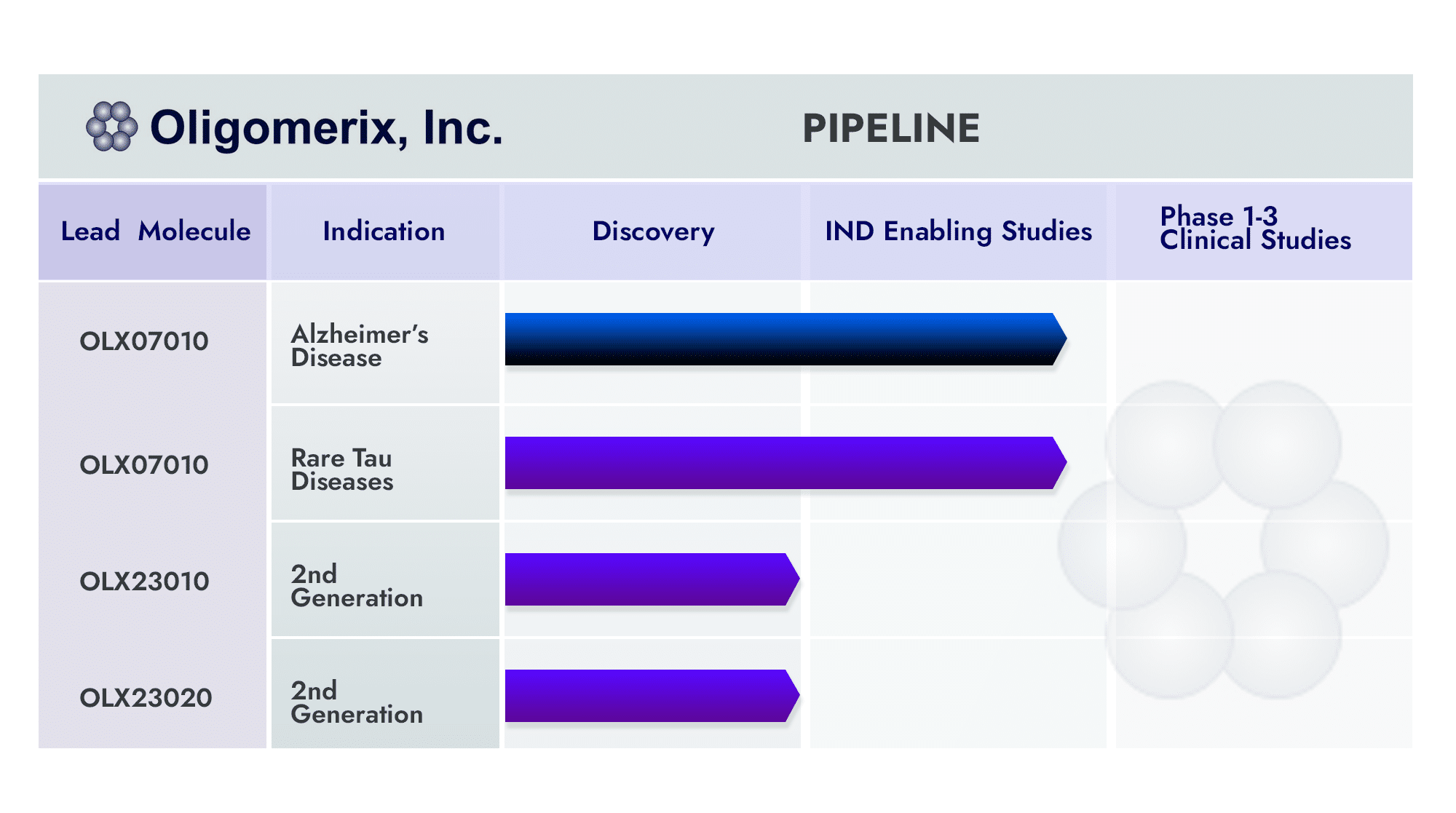
The Company has developed a platform of assays, tools, a lead compound (OLX 07010) and 2nd generation series, and know-how that will enable its strategic partner/acquirer to move rapidly through early and late clinical evaluation, regulatory submission and launch. The hope is to make available treatment options for neurodegenerative disease sufferers and their caregivers for these devastating diseases.
The Need:
There exists a major unmet medical need in the U.S for disease-modifying therapeutics (DMT) for the treatment of dementia in Rare Diseases such as progressive supranuclear palsy (PSP), frontotemporal dementia (FTD), ALS and the more common Alzheimer’s disease (AD). This unmet need continues to grow given both the increasing prevalence and significantly increasing caregiver costs driven by the aging population. For example, death due to AD remains the 6th leading cause of death in the U.S., increasing 145% from 2000 to 2019, and remains as one of the costliest diseases in the U.S. (2023 Alzheimer’s Association Facts and Figures).
Oligomerix is working towards meeting the global need for effective therapeutic strategies. Our oral, small molecule lead program has several potential advantages including:
- Ease of administration to improve patient compliance
- Smaller patient burden for safety monitoring compared with recently approved antibody treatments.
- Low cost of manufacturing for lower cost of therapy
- Increased patient accessibility on a global scale
- Established brain penetration and can also penetrate neurons where tau aggregation is occurring
- Oligomerix’s oral small molecule formulation is a critical competitive advantage because it enables cost-effective, patient-centered treatment for the rapidly growing, unserved AD market in the context of significant and growing global healthcare system cost pressures.
Our platform program is progressing to fill this need with a small molecule tau protein targeted DMT and associated biomarker. According to the World Health Organization, there are around 50 million people who suffer from dementia, and almost 10 million new cases reported every year.
While AD is the most common form of dementia, rare neurodegenerative diseases such as PSP and FTD are still significant healthcare and economic problems. About 30,000 people in the U.S. suffer from PSP (https://www.psp.org/iwanttolearn/progressive-supranuclear-palsy#:~:text=PSP%20is%20considered%20rare.,PSP%20in%20the%20United%20States), while FTD affects 50,000 to 60,000 Americans.
History & Mission:
Origin of the Company: Oligomerix has advanced its inhibitor OLX-07010 tau self-association inhibitor into first-in-human phase 1a clinical trial in early 2023. This achievement is the result of a sole focus on the tau target for more than 18 years, enhancing our understanding of the role that tau plays in AD and other tauopathies such as PSP.
The discovery platform includes a second generation series of small molecules and a companion biomarker program. Oligomerix has advanced its technologies using its equity investment (seed through Series B financing and convertible notes), NIA/NIH grant awards, a grant from the Alzheimer’s Drug Discovery Foundation (ADDF), and tax credits from New York State (NYS) and New York City (NYC). The NIA has been a leading supporter since the Company was founded via its SBIR grant support.
MISSION: Oligomerix seeks to improve the lives of patients with neurodegenerative diseases by discovering and developing a portfolio of disease-modifying, oral small molecule therapeutics that are simple to use.
The Company has built a sound business model around its core technologies, a strong intellectual property (IP) position and a very highly experienced management team.
Oligomerix’s Value Proposition:
Oligomerix discovered and developed a highly innovative and proprietary screening assay that allows it to identify compounds that block the first step in protein aggregation. Utilizing this assay, a diverse library of drug-like compounds was screened, from which emerged our lead candidate OLX-07010. The screening assay, along with OLX-07010 and other second-generation series of compounds, and a focus on lessening the patient burden associated with newer antibody treatments, are the foundation for Oligomerix’s platform and value proposition.
Differentiation: The program is highly differentiated from competitors by targeting tau self-association at the beginning of the tau aggregation cascade that ultimately leads to formation of tau tangles, a hallmark of tau dementias.
Compared to other therapies currently available or in development, our lead program OLX-07010 offers several potential advantages including increased brain penetration, lower cost of manufacturing, ease of oral administration for patient compliance and better patient accessibility on a global scale. Furthermore, OLX-07010 could complement both tau and beta-amyloid antibody treatment.